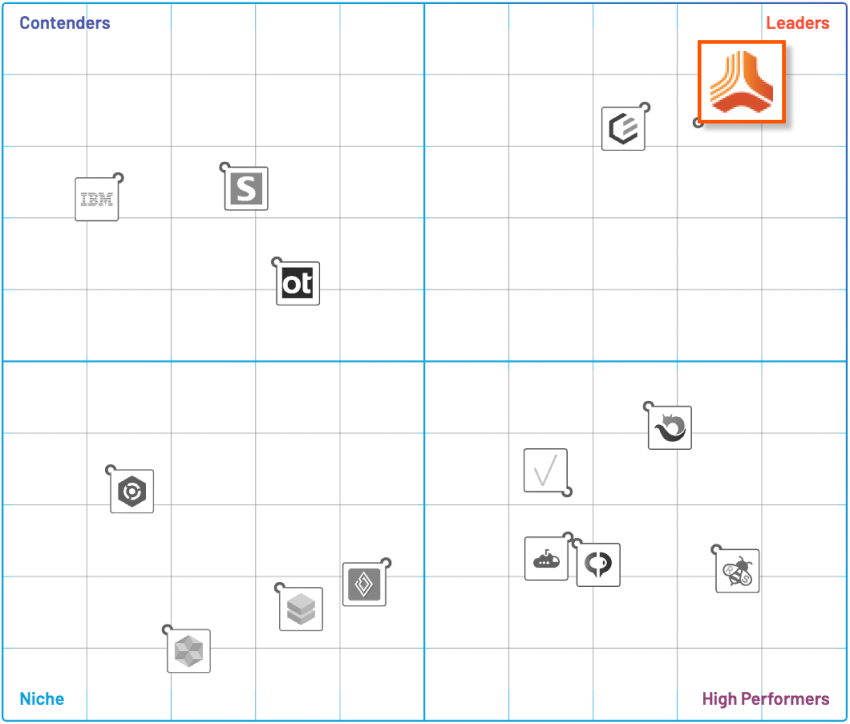
Jama Connect’s Requirements Management Enables Live Traceability™ Across Your Development Process
Bridge engineering silos across development, test, and risk activities. Provide end-to-end compliance, risk mitigation, and process improvement with our intuitive, award-winning requirements management platform.